C'mon river! tell us the difference!!!Hot or cold .
This will be good.
C'mon river! tell us the difference!!!Hot or cold .
C'mon river! tell us the difference!!!
This will be good.
Well? How about you stop being obtuse and avoiding, and answer the question.No . The point when either becomes a liquid . On their own .
The temperature
When hydrogen becomes a liquid .
When oxygen becomes a liquid .
First, neither is a liquid.No . The point when either becomes a liquid . On their own .
When hydrogen becomes a liquid . When oxygen becomes a liquid .
Physical properties:Oxygen is a colourless, odourless, tasteless gas essential to living organisms, being taken up by animals, which convert it to carbon dioxide; plants, in turn, utilize carbon dioxide as a source of carbon and return the oxygen to the atmosphere.
Physical propertiesHydrogen; Hydrogen, H, is a chemical element with atomic number 1. At standard temperature and pressure, hydrogen is a colorless, odorless, nonmetallic, tasteless, highly flammable diatomic gas with the molecular formula H2.
Well? How about you stop being obtuse and avoiding, and answer the question.
You're being obtuse again river...or maybe just stupid.There is nothing obtuse to when oxygen and hydrogen become a liquid . At extremely low temperatures .
you replied...OK, can you tell the difference between a hydrogen atom (H) and an oxygen atom (O) ?
OK, now you have two questions to answer river....what is the difference between an O atom and a H atom.hot or cold?
You're being obtuse again river...or maybe just stupid.
You were asked....
you replied...
OK, now you have two questions to answer river....what is the difference between an O atom and a H atom.
And what difference does being hot or cold make it.
Then you raise being a liquid.
It's obvious you cannot answer the question.
See what happens when you check out a reliable reputable source river? That's science. Good work!Oxygen belong to the group 16 element and hydrogen was the first element in the periodic table. Oxygen contains 8 electrons and eight protons like hydrogen, which has one electron and one proton. ...
See what happens when you check out a reliable reputable source river? That's science. Good work!
Still you are slightly confused. Atomic Oxygen cannot exist in a free state and only consists of one atom, symbol O] while the more commonly referred to molecular Oxygen, [symbol O2] has two atoms and electrons.
Wait, river, in this exercise you are not allowed to cite any mathematical properties, unless you want to prove my point for me.Oxygen belong to the group 16 element and hydrogen was the first element in the periodic table. Oxygen contains 8 electrons and eight protons like hydrogen, which has one electron and one proton. ...
And the ratio of that free oxygen to the rest of the H2O is?So free oxygen in the oceans does not exist
Atomic Oxygen does not exist anywhere in a free state.So free oxygen in the oceans does not exist .
river said: ↑
Oxygen belong to the group 16 element and hydrogen was the first element in the periodic table. Oxygen contains 8 electrons and eight protons like hydrogen, which has one electron and one proton. ...
Wait, river, in this exercise you are not allowed to cite any mathematical properties, unless you want to prove my point for me. It's the mathematical values that determine the state or dynamics of any and all objects and systems (patterns).
Of course! even if you did need to google it from a reputable source. We all do that river, or at least those interested in science do.I am allowed to state their Physical Properties .
Atomic Oxygen does not exist anywhere in a free state.
Of course you are, but that does not have any practical meaning. The universe is a very practical place. Our science is based on its consistency of functional interactions.I am allowed to state their Physical Properties .
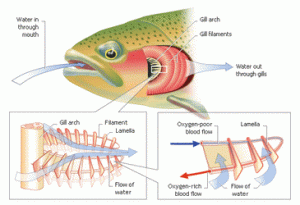
Even though fish can live their lives underwater, they still need oxygen to “breathe”. Instead of breathing air, fish must get their oxygen from the water. This process requires large volumes of water to pass through absorption surfaces to get enough oxygen into their bodies using their mouths and gills. These body parts work like a pump to keep water moving over the gas absorption surfaces of the gills.
Surface area is always the answer to most science problems! The more surface area fish have on the gills, the easier they will be able to absorb oxygen. If the skin on the gills is thin, this also helps. The blood passing through the gills is pumped in the opposite direction of the water flowing over the gills. This allows the blood oxygen level to be less than the oxygen level in the water. Oxygen will always want to move to wherever lacks oxygen, this is also known as diffusion. The oxygen can then attach to hemoglobin, a protein in red blood cells that distributes oxygen throughout the body.
Nup, I'm correct river. You simply are confusing atomic and molecular Oxygen...or O and O2You are wrong .
Think of fish , they breath free oxygen .
Think pad , aquariums, they need oxygenation .
Of course you are, but that does not have any practical meaning. The universe is a very practical place. Our science is based on its consistency of functional interactions.
OK, and what are the relational values of these physical properties? Else how do you know the difference between oxygen an hydrogen?
Nup, I'm correct river. You simply are confusing atomic and molecular Oxygen...or O and O2
Or are you going to keep making a fool of yourself as you did with your claim of the Moon not rotating and having one side facing Earth. We all,breath molecular Oxygen river.
https://www.nasa.gov/topics/technology/features/atomic_oxygen.html
Understanding Atomic Oxygen.
::::::::::::::::::::::::::::::::::::::::::::::::::::
Oxygen comes in several different forms. The oxygen that we breathe is called O2—that is, it is comprised of two atoms of oxygen. O3 is ozone, such as occurs in Earth's upper atmosphere, and O (one atom), is atomic oxygen.
Atomic oxygen doesn't exist naturally for very long on the surface of Earth, as it is very reactive. But in space, where there is plenty of ultraviolet radiation, O2 molecules are more easily broken apart to create atomic oxygen. The atmosphere in low Earth orbit is comprised of about 96% atomic oxygen. In the early days of NASA's space shuttle missions, the presence of atomic oxygen caused problems.
"In the first few shuttle flights, materials looked frosty because they were actually being eroded and textured," says Bruce Banks, a senior physicist with Alphaport, supporting the Space Environment and Experiments branch at Glenn. "Atomic oxygen reacts with organic materials on spacecraft exteriors, gradually damaging them."
Glenn Research Center was asked to investigate the damage caused to NASA spacecraft by atomic oxygen. The researchers not only invented methods to protect spacecraft from atomic oxygen; they also discovered a way to harness the potentially destructive power of atomic oxygen and use it to improve life on Earth.
You didn't answer my question . What oxygen do Fish breath in .
Fish of course extract the Oxygen from the water, but as per the atmosphere, the free atomic Oxygen does not exist other then as the covalent bond with Hydrogen.
