Hi Andrew,A quick search on the web revealed these about dark skin:
www.modelofreality.org/UV1.gif
www.modelofreality.org/UV2.gif
(be sure to zoom in if necessary).
I'll see your random websites and raise you some real references.
K.F. KÖLMEL, B. SENNHENN, K. GIESE (1990), Investigation of skin by ultraviolet remittance spectroscopy, British Journal of Dermatology 122 (2), 209–216.:
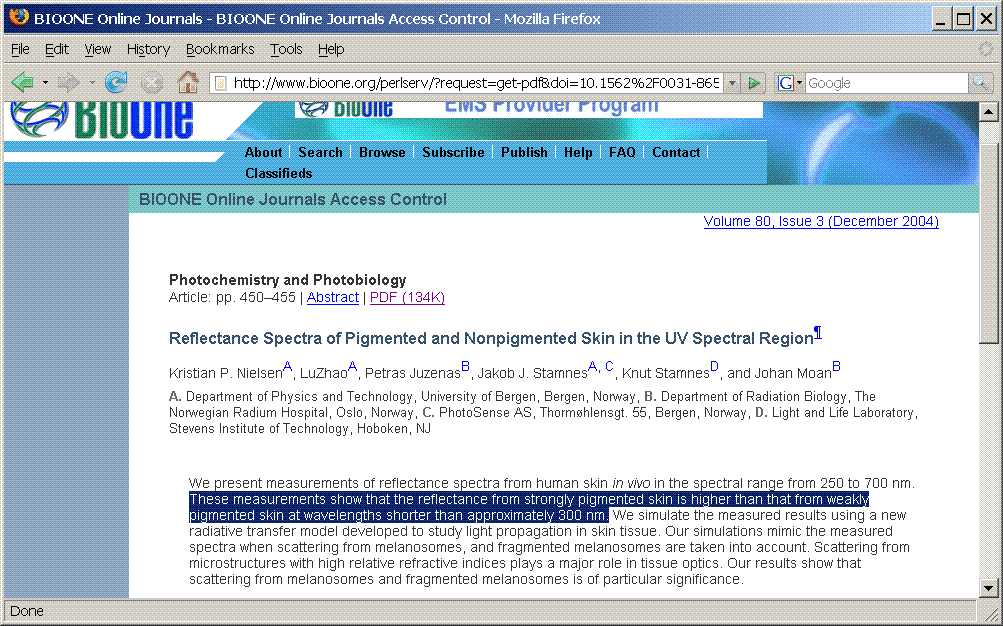
"The increase in melanin content... lowers the remittance at all UV wavelengths"
Angelopoulou, Elli, 1999, The Reflectance Spectrum of Human Skin, Technical Report MS-CIS-99-29, GRASP Laboratory, Department of Computer and Information Science, University of Pennsylvania.
http://www.cis.upenn.edu/~elli/tech-report.skin.pdf
"In general, the reflectance spectrum of melanin in the visible range is monotonically increasing with wavelength with maximum absorption occurring in the UV range. Buckley and Grum [1] who compared non-pigmented versus healthy adjacent patches of skin in vitiligo patients observed a similar melanin reflectance curve."
Tadokoro, T, Kobayishi, N, Zmudzka, B, Ito, S, Wakamatsu, K, Yamaguchi, Y, Korossy, K, Miller, S, Beer, J, and Hearing, V, UV-induced DNA damage and melanin content in human skin differing in racial/ethnic origin, FASEB J. 2003 17: 1177-1179.
http://www.fasebj.org/cgi/reprint/17/9/1177.pdf
"Melanin can absorb UV efficiently at most wavelengths"
The UV is absorbed by the melanin, which prevents it reaching the skin cells. The difference isn't whether the UV is absorbed, it's what absorbs it.But it only makes sense. UV absorption is responsible for Vitamin D production.
So Blacks get rickets in Northern Europe since they reflect UV radiation.
Blacks don't sunburn so easily near the equator. That's because they reflect the damaging UV "rays".
I'd be pretty surprised if fit hasn't been done. UV spectroscopy isn't that hard, is it? Have you done a thorough search of the literature? Have you asked someone in that field?Now Lampblack. I actually wanted to measure the UV reflectivity of this material in the UT physics lab, but as I said earlier, it did not work out. So this experiment is one which I hope someone will do very soon.
This is one of my suggested "New Experiments".
Try someone in materials science. Carbon black is used in paint pigments, and UV reflectance of paints is a health issue.
This rather large pdf from a paint manufacturer says:
Durability - The ability of the paint film to stand up to the ravages of exposure to the elements and physical abuse is a measure of durability. Carbon black’s excellent absorber of all wavelengths of light make it a good choice for providing UV protection for the paint system it is dispersed in. Finer blacks tend to absorb more UV than coarser ones, thus finer blacks are preferred where UV protection is required. Increased loadings also result in increased UV absorption."
Here's something else of interest. Not specifically about lampblack, but at least it has measurements:
Why do you think it's certain? Do you suggest that all absorbed radiation causes degradation in all materials?Lampblack seems to be well known for its "UV stabilizing" properties.
UV resistance makes one think that Lampblack is a UV reflector, right?
If it was an absorber of UV, degradation of the material would certainly occur. Hmmm. . .
I think that carbon black is good at protecting materials from UV for the same reason that melanin is good at protecting skin cells.
Didn't you get enough of that at PhysicsForums?I want to hear all of your objections.
You're obviously a smart and well-educated guy, Andrew... but I have the suspicion that maybe you've stepped beyond your field of expertise, and are blinded by your own brilliance.
Be honest now... have you seriously tried to find out whether melanin and carbon black are good UV reflectors? Or have you only looked for information that support your pet theory?
Last edited: