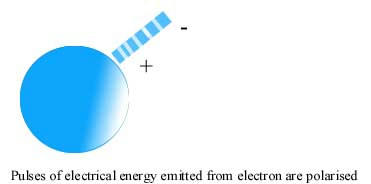
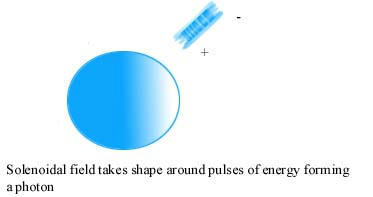
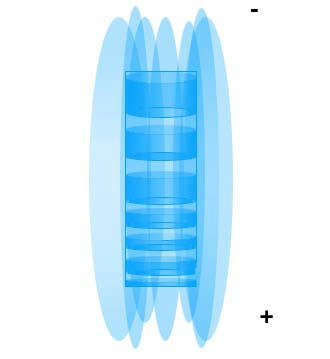
A Gestalt Theory of the nature of light and related phenomenon:
The main drawback with QM is that it has established certain inviolable tenets or beliefs that, as long they are open to other explanations , are absolutely self defeating and have no place in a science that claims to be based on pure reason and a detached and neutral point of view. For instance one of the reasons that QM puts forward for the inadequacy of classical physics is the fact, that using purely classical physical theories ‘atoms cannot exist !’ Absurd as it sounds, a lot of the hype and baggage that QM has collected over the years, is based on the facts that classical physics has no explanation for how ‘atoms can exist.’ According to QM, it follows that the whole edifice on which Classical physics is built collapses at this point and can have no further application in explaining events at the sub-atomic level. However, anyone with even a modicum of common sense, can see at once that this is a childish argument, because no time or latitude is given for Classical Physics to come up with an explanation. In saner times, what would have been said was that classical theories do not at present appear to have an explanation as to how atoms can exist, and until they come up with some explanation the theories put forward by QM will have to be followed as the most likely explanation.
Why can’t atoms exist according to the classical physics point of view: A positively charged nucleus ‘holds’ a negatively charged electron by the force of Coulomb attraction:
1)
$$ F{_coul}= \frac{e^2}{a^2}$$
( e is the electron’s and proton’s charge, a is the radius of an atom, in this case the hydrogen atom). For the atom to be stable, the force of attraction is insufficient and the electron would fall into the nucleus. Therefore this force must be balanced by a corresponding force of repulsion, which is supplied by centrifugal force:
2)
$$F_c = \frac{mv^2}{a}$$
Here m is the electron’s mass and v is its velocity. The equilibrium of forces makes it possible to determine the velocity of the electron in its orbit:
3)
$$ v = (\frac{e^2}{ma})^\frac{1}{2}$$
Substituting the numerical values for charge, the electron’s mass, and the radius of the atom
(3 x $$ 10^ {-8} $$cms approx) yields the answer :$$ v \approx 10^ {8}$$ cms/sec or 1000 kmh.
Next we need to know the total energy of the electron in the field of the nucleus:
4)
$$E = -\frac{e^2}{2a}$$
This formula is obtained by the summation of the kinetic energy and the potential energy of the electron. Finally, the radiation intensity of a charge e moving with acceleration w can be calculated : $$ w = \frac{v^2}{a} $$
5)
$$I = \frac{2e^2w^2}{3c^2} = \frac {2e^2v^4}{3c^3a^2}$$ ergs/sec
If the electron emits I ergs each second, then it will lose all its energy in a time interval:
6)
$$T\approx\frac{a}{v}(\frac{c}{v})^2$$
Using this equation it is possible to calculate that $$ T \approx 10 ^{-10} $$ Sec. Thus according to Classical physics , the electron should fall into the nucleus in about $$10^{-10}$$ secs and therefore atoms cannot exist. So far so good, it has been adequately demonstrated that the existing classical physics explanations for the phenomenon are inadequate. Yet QM tends to frame this inability in terms of absolutes with statements such as : “This is the best demonstration of the total failure of Classical Physics.” And “This is possibly the most impressive contradiction between classical physics and experiment and so on. “The new mechanics was developed precisely to eliminate this contradiction.” Impressive, but what was the QM explanation of the phenomenon ?
New information on the structure of the atom was emerging so fast at this time in the 1920’s that there was hardly time to assimilate it. Yet the proponents of the wave theory seemed to have the upper hand, precisely because of seemingly insoluble problems such as that of the existence of the atom referred to earlier. Obviously a wave is not localized, if the electron was considered as a wave the problem of the ‘radiating electron falling into the nucleus’ went away since the electron was spread out and not localized. This theory seemed to gain ground with Louis De Broglie’s theory of matter waves, since waves were associated with matter it seemed obvious that waves could also be associated with electrons. The only problem with this theory, as with so many other theories in Quantum Mechanics, was that neither De Broglie, nor anyone else could explain what was waving. To this day more than seventy years after De Broglie formulated his theory, no-one can come up with an explanation of what exactly is supposed to be waving when one speaks of matter waves, or waves associated with matter. The German physicist Erwin Schrodinger thought that he had solved the problem when he described electrons as being standing waves. Again, as so often happens in the history of QM, Schrodinger was not content with giving a description of electrons where their motion or position was described in terms of standing waves, he went much further by stating that electrons were actually standing waves. He compiled numerous diagrams showing the shape that ‘standing wave’ electrons might take. Here is an example.

Other physicists elaborated or rather generalized on this theory by stating that the electron was a 'cloud'. The Schrodinger wave theory was at first wildly successful, physics text books in schools and colleges were altered to state that electrons travelled round the nucleus as a cloud rather than as a point particle as Bohr’s planetary model of the atom had implied. Soon, however, cracks began to show in Schrodinger’s theory, it is a little known fact that Schrodinger’s wave theory although sufficient to describe the simplest of atoms grew in complexity when it came to describing larger atoms, they became so complex in fact that to describe the Uranium atom required, no less than 276 dimensions. Don’t even ask me what this means because quite frankly it is beyond comprehensions. To try and comprehend the 4 or 5 dimensions of Brane Theory or the 11 dimensions put forward by String Theory is befuddling enough, but 276 dimensions is really out of this world the Universe and everything else. Obviously when Schrodinger came to this conclusion and admitted that his theory was untenable. Schools and Colleges once again altered their text books to delete any references to electrons as clouds. What happens next is even more bizarre. The Danish scientist Neils Bohr had been doing some incredible work improving his model of the atom by working on the problem of spectral lines. Scientists at this time could not understand how a simple Hydrogen atom , having only one electron could emit a spectrum that had over a hundred lines. Bohr proved that when electrons absorbed an electron they would jump out to an outer orbit and then emit the same amount of energy as they moved back to their original position. In this way Bohr was able to explain, how many electrons each orbit could contain, and how much energy would be released when an electron ‘jumped’ from one orbit to another. This was a truly incredible piece of work based on the collation of empirical data collected from experiments. In that sense it was a purely classical investigation. It explained every element in the periodic table, the number of electrons in its shells, its physical properties and the spectrum it radiated .When Werner Heisenberg, who had been working on the same problems as Schrodinger, learnt that Schrodinger was having problems and was about to renounce his wave theory, he convinced him not to renounce his theory. He explained to Schrodinger that instead of using the mathematical formula which was extremely bulky and complicated, he had devised a matrix look up table that would give exactly the same result. In actual fact all that Heisenberg had done was to tabulate Bohr’s results in the form of look up matrix tables. These tables called the s-matrix are still being used today. Of course in the intervening years the s-matrix has undergone many changes and improvements, in this particular instance, I am referring to the matrix prepared by Heisenberg that gave the probability from look up tables, of the number of electrons in each shell of the an atom, which was what Schrodinger had set out to solve with his wave function. The irony of the situation is that data compiled through meticulous research conducted on the lines of classical physics was used to shore up an absurd theory. (Like it or not, any theory which requires 276 dimensions to work is patently absurd and completely unacceptable.) The German physicist Max Born also came to Schrodinger’s aid by suggesting that the wave function did not refer to any practical object but rather was a manifestation of probabilities. Still, a probability function that requires 276 dimensions, it’s a bit much. However, there is no doubt that the famous Schrodinger wave function equation, remains to this day the single most important mathematical element in quantum mechanics. It is described as being a composite of all the possibilities of the system being observed. Using the Schrodinger wave function it is claimed that quantum theory can predict all the probability of a microscopic event with the same precision that Newtonian physics can predict the actual occurrence of a macroscopic event.
Contd...........
The main drawback with QM is that it has established certain inviolable tenets or beliefs that, as long they are open to other explanations , are absolutely self defeating and have no place in a science that claims to be based on pure reason and a detached and neutral point of view. For instance one of the reasons that QM puts forward for the inadequacy of classical physics is the fact, that using purely classical physical theories ‘atoms cannot exist !’ Absurd as it sounds, a lot of the hype and baggage that QM has collected over the years, is based on the facts that classical physics has no explanation for how ‘atoms can exist.’ According to QM, it follows that the whole edifice on which Classical physics is built collapses at this point and can have no further application in explaining events at the sub-atomic level. However, anyone with even a modicum of common sense, can see at once that this is a childish argument, because no time or latitude is given for Classical Physics to come up with an explanation. In saner times, what would have been said was that classical theories do not at present appear to have an explanation as to how atoms can exist, and until they come up with some explanation the theories put forward by QM will have to be followed as the most likely explanation.
Why can’t atoms exist according to the classical physics point of view: A positively charged nucleus ‘holds’ a negatively charged electron by the force of Coulomb attraction:
1)
$$ F{_coul}= \frac{e^2}{a^2}$$
( e is the electron’s and proton’s charge, a is the radius of an atom, in this case the hydrogen atom). For the atom to be stable, the force of attraction is insufficient and the electron would fall into the nucleus. Therefore this force must be balanced by a corresponding force of repulsion, which is supplied by centrifugal force:
2)
$$F_c = \frac{mv^2}{a}$$
Here m is the electron’s mass and v is its velocity. The equilibrium of forces makes it possible to determine the velocity of the electron in its orbit:
3)
$$ v = (\frac{e^2}{ma})^\frac{1}{2}$$
Substituting the numerical values for charge, the electron’s mass, and the radius of the atom
(3 x $$ 10^ {-8} $$cms approx) yields the answer :$$ v \approx 10^ {8}$$ cms/sec or 1000 kmh.
Next we need to know the total energy of the electron in the field of the nucleus:
4)
$$E = -\frac{e^2}{2a}$$
This formula is obtained by the summation of the kinetic energy and the potential energy of the electron. Finally, the radiation intensity of a charge e moving with acceleration w can be calculated : $$ w = \frac{v^2}{a} $$
5)
$$I = \frac{2e^2w^2}{3c^2} = \frac {2e^2v^4}{3c^3a^2}$$ ergs/sec
If the electron emits I ergs each second, then it will lose all its energy in a time interval:
6)
$$T\approx\frac{a}{v}(\frac{c}{v})^2$$
Using this equation it is possible to calculate that $$ T \approx 10 ^{-10} $$ Sec. Thus according to Classical physics , the electron should fall into the nucleus in about $$10^{-10}$$ secs and therefore atoms cannot exist. So far so good, it has been adequately demonstrated that the existing classical physics explanations for the phenomenon are inadequate. Yet QM tends to frame this inability in terms of absolutes with statements such as : “This is the best demonstration of the total failure of Classical Physics.” And “This is possibly the most impressive contradiction between classical physics and experiment and so on. “The new mechanics was developed precisely to eliminate this contradiction.” Impressive, but what was the QM explanation of the phenomenon ?
New information on the structure of the atom was emerging so fast at this time in the 1920’s that there was hardly time to assimilate it. Yet the proponents of the wave theory seemed to have the upper hand, precisely because of seemingly insoluble problems such as that of the existence of the atom referred to earlier. Obviously a wave is not localized, if the electron was considered as a wave the problem of the ‘radiating electron falling into the nucleus’ went away since the electron was spread out and not localized. This theory seemed to gain ground with Louis De Broglie’s theory of matter waves, since waves were associated with matter it seemed obvious that waves could also be associated with electrons. The only problem with this theory, as with so many other theories in Quantum Mechanics, was that neither De Broglie, nor anyone else could explain what was waving. To this day more than seventy years after De Broglie formulated his theory, no-one can come up with an explanation of what exactly is supposed to be waving when one speaks of matter waves, or waves associated with matter. The German physicist Erwin Schrodinger thought that he had solved the problem when he described electrons as being standing waves. Again, as so often happens in the history of QM, Schrodinger was not content with giving a description of electrons where their motion or position was described in terms of standing waves, he went much further by stating that electrons were actually standing waves. He compiled numerous diagrams showing the shape that ‘standing wave’ electrons might take. Here is an example.

Other physicists elaborated or rather generalized on this theory by stating that the electron was a 'cloud'. The Schrodinger wave theory was at first wildly successful, physics text books in schools and colleges were altered to state that electrons travelled round the nucleus as a cloud rather than as a point particle as Bohr’s planetary model of the atom had implied. Soon, however, cracks began to show in Schrodinger’s theory, it is a little known fact that Schrodinger’s wave theory although sufficient to describe the simplest of atoms grew in complexity when it came to describing larger atoms, they became so complex in fact that to describe the Uranium atom required, no less than 276 dimensions. Don’t even ask me what this means because quite frankly it is beyond comprehensions. To try and comprehend the 4 or 5 dimensions of Brane Theory or the 11 dimensions put forward by String Theory is befuddling enough, but 276 dimensions is really out of this world the Universe and everything else. Obviously when Schrodinger came to this conclusion and admitted that his theory was untenable. Schools and Colleges once again altered their text books to delete any references to electrons as clouds. What happens next is even more bizarre. The Danish scientist Neils Bohr had been doing some incredible work improving his model of the atom by working on the problem of spectral lines. Scientists at this time could not understand how a simple Hydrogen atom , having only one electron could emit a spectrum that had over a hundred lines. Bohr proved that when electrons absorbed an electron they would jump out to an outer orbit and then emit the same amount of energy as they moved back to their original position. In this way Bohr was able to explain, how many electrons each orbit could contain, and how much energy would be released when an electron ‘jumped’ from one orbit to another. This was a truly incredible piece of work based on the collation of empirical data collected from experiments. In that sense it was a purely classical investigation. It explained every element in the periodic table, the number of electrons in its shells, its physical properties and the spectrum it radiated .When Werner Heisenberg, who had been working on the same problems as Schrodinger, learnt that Schrodinger was having problems and was about to renounce his wave theory, he convinced him not to renounce his theory. He explained to Schrodinger that instead of using the mathematical formula which was extremely bulky and complicated, he had devised a matrix look up table that would give exactly the same result. In actual fact all that Heisenberg had done was to tabulate Bohr’s results in the form of look up matrix tables. These tables called the s-matrix are still being used today. Of course in the intervening years the s-matrix has undergone many changes and improvements, in this particular instance, I am referring to the matrix prepared by Heisenberg that gave the probability from look up tables, of the number of electrons in each shell of the an atom, which was what Schrodinger had set out to solve with his wave function. The irony of the situation is that data compiled through meticulous research conducted on the lines of classical physics was used to shore up an absurd theory. (Like it or not, any theory which requires 276 dimensions to work is patently absurd and completely unacceptable.) The German physicist Max Born also came to Schrodinger’s aid by suggesting that the wave function did not refer to any practical object but rather was a manifestation of probabilities. Still, a probability function that requires 276 dimensions, it’s a bit much. However, there is no doubt that the famous Schrodinger wave function equation, remains to this day the single most important mathematical element in quantum mechanics. It is described as being a composite of all the possibilities of the system being observed. Using the Schrodinger wave function it is claimed that quantum theory can predict all the probability of a microscopic event with the same precision that Newtonian physics can predict the actual occurrence of a macroscopic event.
Contd...........